

















QIAGEN Clinical Insight (QCI™) for Somatic Cancer is an integrated clinical decision support solution designed specifically for routine somatic cancer testing laboratories assessing next-generation sequencing (NGS) data. With QCI you can annotate, interpret and report NGS variants in the context of over 10 million relevant biomedical findings while building your own internal experiential knowledge base. QCI allows you to minimize complexity, time, and cost associated with determining clinical significance and action-ability of NGS variants.
QCI Interpret replaces your complex and tedious clinical NGS variant research with a scalable interpretation workflow alternative that helps grow your indication menu and volume. With QCI Interpret you can:
Reduce the time, costs, and complexity associated with NGS tests.
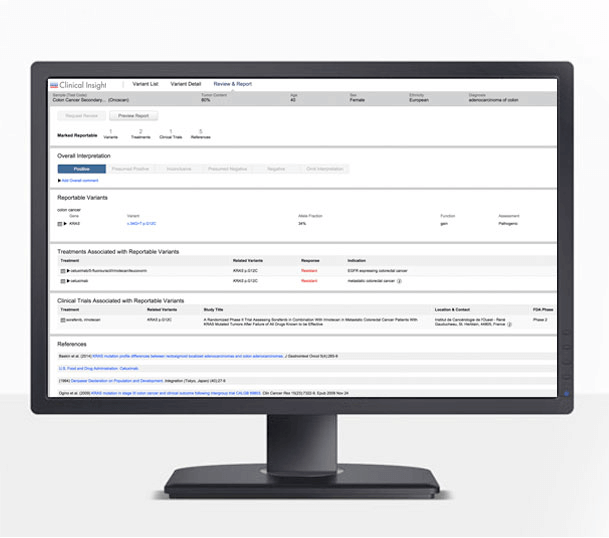
Focus on actionable variants, including co-occurring alterations, to increase treatment and clinical-trial matching.
Build your own private, experience-based database with each variant assessed and reported, increasing speed and accuracy with subsequent interpretations.

QCI is an interpretation productivity tool that helps identify actionable variants fast with results you can trust, all in one secure web application. With QCI Interpret you get:

*QCI Interpret is an evidence-based decision support software intended as an aid in the interpretation of variants observed in genomic sequencing data. The software evaluates genomic variants in the context of published biomedical literature, professional association guidelines, publicly available databases and annotations, drug labels, and clinical-trials. Based on this evaluation, the software proposes a classification to aid in the interpretation of observed variants. The software is NOT intended as a primary diagnostic tool by physicians or to be used as a substitute for professional healthcare advice. Each laboratory is responsible for ensuring compliance with applicable international, national, and local clinical laboratory regulations and other specific accreditations requirements.