


















Recognize rare mutations of cardiomyopathies with database and software solutions for analyzing and interpreting genetic abnormalities of inherited cardiovascular diseases
Inherited cardiomyopathies (ICs) are a major cause of heart disease and progressive heart failure, affecting approximately 1 in 500 individuals (1). Consequently, genetic testing is used to confirm a diagnosis, guide management, and provide disease prognosis.
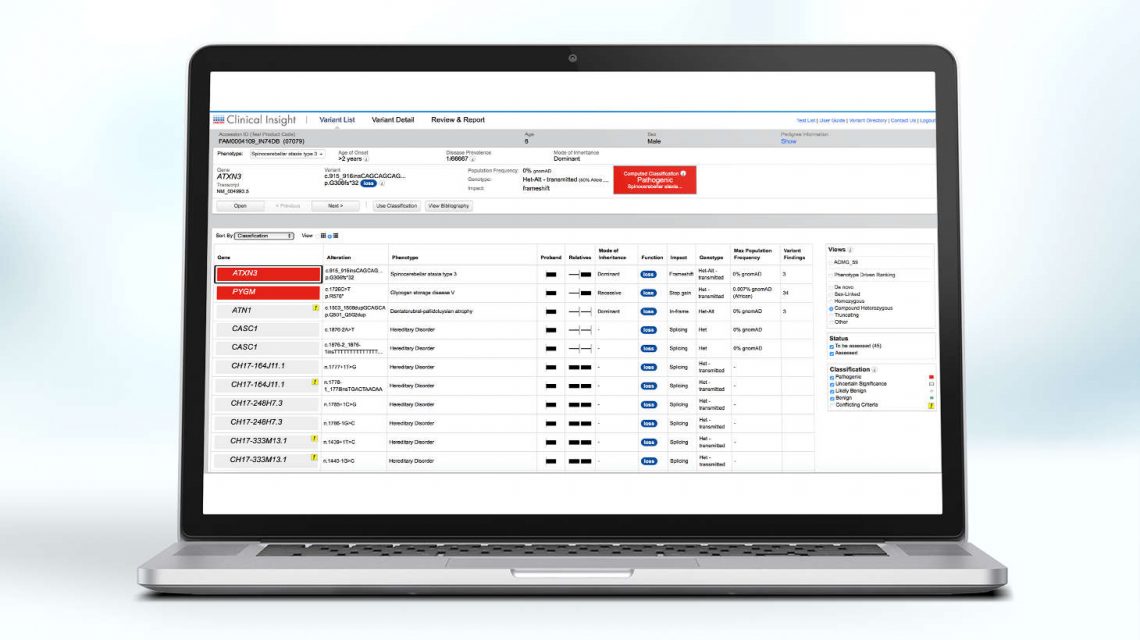
NGS-based assays for the rapid and cost-effective molecular diagnosis of diverse ICs are becoming a routine part of clinical care. While larger, multigene panels offer higher diagnostic yields, there is a concurrent increase in the rate of inconclusive results, illuminating the importance of high-quality and confident variant interpretation.
Inherited cardiomyopathies are heterogeneous disorders of the myocardium formally classified into six distinct forms:
Approximately 60 genes have been reported to be disease-related in inherited cardiomyopathy (2). While large multigene NGS panels provide opportunities to investigate thousands of genetic aberrations simultaneously among diverse cardiomyopathy phenotypes, identifying clinically actionable variants remains a challenge.
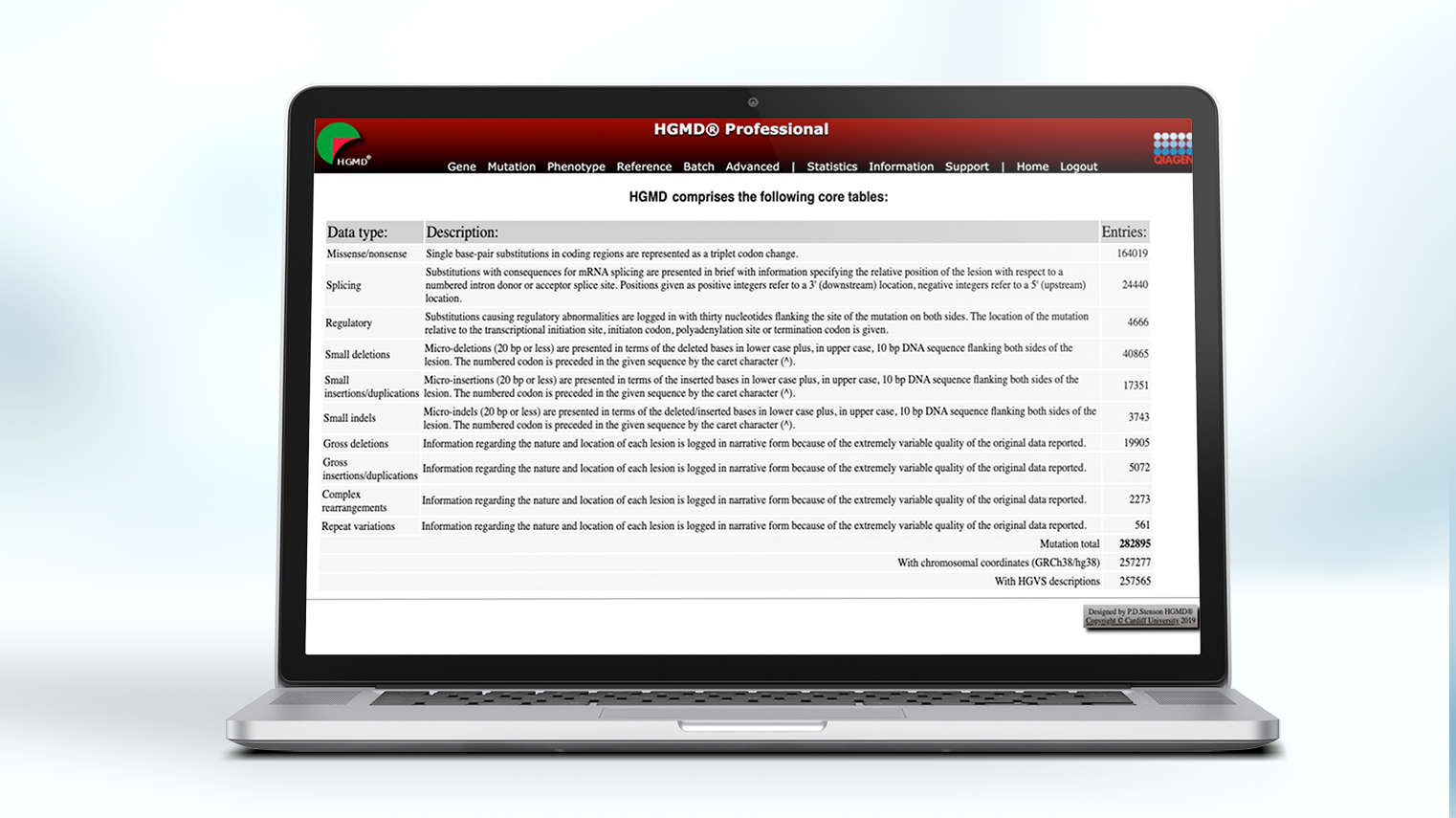
The content core of QIAGEN's clinical NGS variant interpretation and reporting solutions, the QIAGEN Knowledge Base (QKB) is unrivalled in breadth, depth, and accuracy, ensuring labs have access to the evidence, peer-reviewed literature, guidelines, drug labels, and clinical trial registries needed to interpret every variant with precision and confidence.
Built over 20 years and encompasses over 40 public and proprietary databases
Maintained by hundreds of certified MD- and PhD-level expert curators who enter more than 5,000 new findings each day
Contains the latest evidence from peer-reviewed papers, clinical and functional studies, on- and off-label drugs, and professional guidelines, such as NCCN, AMP/ASCO/CAP and the World Health Organization (WHO)
Updates list of recruiting clinical trials each week



References